In July of 1987, as described by U.S. Department of Agriculture researchers Idso and Kimball (2001), eight 30-cm-tall sour orange tree (Citrus aurantium L.) seedlings were planted directly into the ground at the Agricultural Research Service's U.S. Water Conservation Laboratory in Phoenix, Arizona, where they were enclosed in pairs within four clear-plastic-wall open-top chambers. Then, in November of that year, the two scientists began to continuously pump ambient air through two of the chambers via perforated plastic tubes that lay upon the ground beneath the trees, while through the other two chambers they began to pump air that was enriched with carbon dioxide to a concentration that was 300 ppm greater than that of the surrounding ambient air, which had an average CO2 concentration of 400 ppm. And thus was born one of the longest atmospheric CO2 experiments ever to be conducted anywhere in the world.
Throughout the experiment, the Phoenix global-change research team irrigated and fertilized the trees according to standard agronomic practices in the area, in order to keep them as free as possible from water and nutrient stresses; and they measured the circumferences of the trees' trunks at a height of 45 cm above the surface of the ground at the midpoint of every month. At the ends of the second and third years of the study, they also determined the total trunk and branch volume of each tree from trunk and branch length and diameter measurements; and from these data they developed a relationship between trunk cross-sectional area and trunk plus branch volume that applied equally well to both the CO2-enriched and ambient-treatment trees. Then they made numerous wood density measurements that finally allowed them to calculate the total aboveground woody biomass of each tree at the midpoint of every month. In addition, all of the oranges produced by the trees were picked, counted and weighed each year; and a large number of the fruit were dried in ovens to determine the amount of dry matter they contained. And by these means the two researchers developed a yearly record of total fruit biomass production to accompany their monthly record of wood biomass production.
As the experiment progressed, the CO2-enriched/ambient-treatment ratio of cumulative aboveground wood biomass rose rapidly from an initial value of unity to a value slightly in excess of 3.0 at the two-year point of the study, as shown in the figure below, which depicts the changing ratio of the aboveground wood biomass of the CO2-enriched trees to that of the ambient-treatment trees over the first two years of the study.
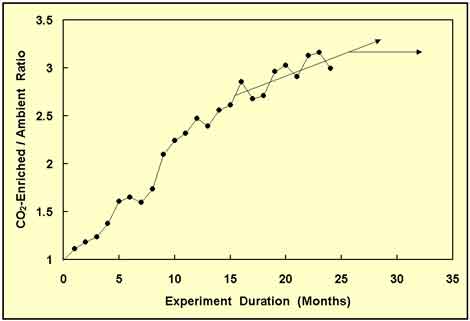
Figure 1. The CO2-enriched/ambient-treatment ratio of total aboveground sour orange tree wood biomass plotted as a function of time since the start of the experiment, along with arrows that depict what would seem to be two logical projections of what might happen to this ratio if the experiment were to be continued beyond the last point shown. Adapted from Idso and Kimball (2001).
From the data of Figure 1, it could reasonably be assumed that the CO2-enriched/ambient-treatment wood biomass ratio might possibly rise a little higher, but that it had to level off sometime soon. Hence, it was decided to continue collecting data a little longer, in order to determine the long-term asymptotic growth response of the trees; and after seven additional months, the results depicted in Figure 2 were obtained.
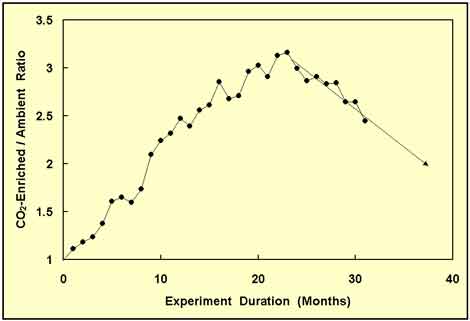
Figure 2. Same as Figure 1, but with seven additional months of data and a new forward projection of the CO2-enriched/ambient-treatment wood biomass ratio. Adapted from Idso and Kimball (2001).
To the researchers' surprise, the wood biomass ratio not only did not rise any higher, or even level out, it began a steep decline. And projecting forward in time, it appeared that if the new trend continued, all of the biomass advantage acquired by the CO2-enriched trees might well disappear over the next couple of years. Clearly, therefore, they had no recourse but to continue the experiment, which they did, obtaining the additional data depicted in Figure 3.
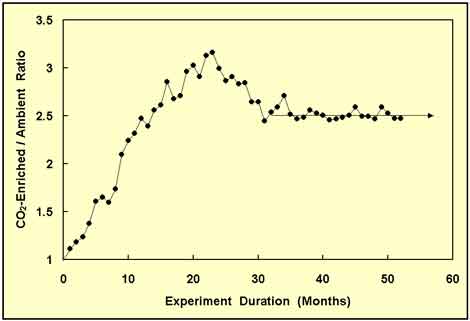
Figure 3. Same as Figure 2, but with 21 additional months of data and a new forward projection of the wood biomass ratio. Adapted from Idso and Kimball (2001).
The results portrayed in Figure 3 were extremely gratifying. They suggested that after an initial "overshoot" of the long-term response of the trees to atmospheric CO2 enrichment, things had finally settled down and a final answer had been obtained: the ultimate equilibrium response of the trees was a whopping 150% increase in wood biomass production in response to the 75% increase in the air's CO2 concentration. For some reason, however, the research team kept on collecting data, only to be surprised once again, as depicted in Figure 4.
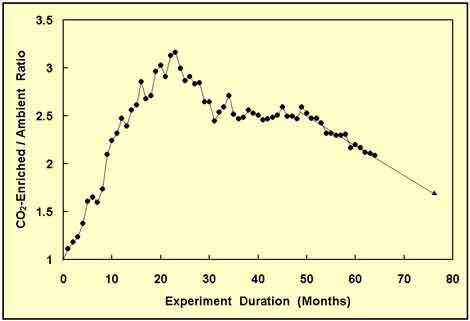
Figure 4. Same as Figure 3, but with 12 more months of data and yet another forward projection of the CO2-enriched/ambient-treatment wood biomass ratio. Adapted from Idso and Kimball (2001).
After having maintained a near-constant value for over a year and a half, the wood biomass ratio began to decline again, as shown in Figure 4, not as rapidly as it had the first time, but at a still substantial rate. And as it dropped ever lower, it was decided to continue collecting data, which yielded the results depicted in Figure 5.
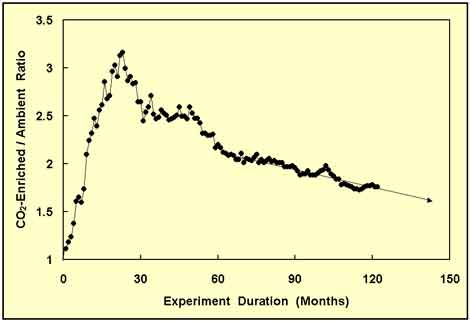
Figure 5. Same as Figure 4, but with 58 months of additional data and one more projection of the CO2-enriched/ambient-treatment wood biomass ratio. Adapted from Idso and Kimball (2001).
This time, the change was to a reduced rate of decline in the wood biomass ratio; and it persisted for nearly five years. At this point, the experiment had passed the decade point; and it looked like the wood biomass ratio was going to continue to slowly decline and probably approach some asymptotic limit that might not be much greater than unity. And as that possibility would be of extreme significance, Idso and Kimball continued collecting data, obtaining the results portrayed in Figure 6.
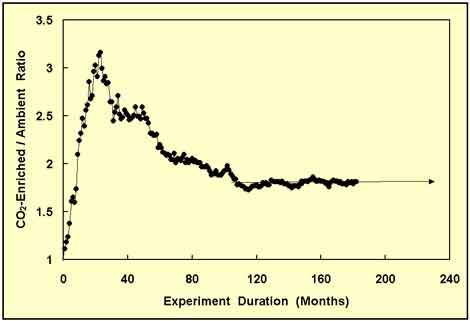
Figure 6. Same as Figure 5, but with 60-plus months of additional data and one last forward projection of the wood biomass ratio. Adapted from Idso and Kimball (2001).
Finally, at the nine-year point of the study, the CO2-enriched/ambient-treatment wood biomass ratio leveled out at a value of 1.80, which value was subsequently assumed to be the true long-term growth response of the trees to the 75% increase in atmospheric CO2 concentration.
Providing additional support for this conclusion, fruit production began in the third year of the study, when the CO2-enriched trees produced an average of 25 fruit per tree and the ambient-treatment trees produced an average of but one fruit per tree. Thereafter, the cumulative CO2-enriched/ambient-treatment fruit biomass ratio also dropped substantially, ultimately leveling out just a little above the CO2-enriched/ambient-treatment wood biomass ratio. And these findings, in the words of Idso and Kimball (2001), "are indicative of the likelihood that the CO2-enriched trees may have reached an equilibrium condition with respect to the CO2-induced enhancement of wood biomass and fruit production, and that they will not substantially depart from these steady-state responses over the remainder of their lifespan."
Giving added confidence to the above conclusions was the ancillary study of Leavitt et al. (2003), who evaluated the intrinsic water use efficiencies of the trees via analyses of the stable carbon isotopes of leaves that had been collected from each of them every two months throughout 1992, as well as on three occasions in 1994-95, plus wood samples that were extracted five years later from two cores that passed through the centers of each of the trees' trunks perpendicular to each other at a height of 45 cm above the ground. The ultimate finding of this endeavor was an 80% increase in intrinsic water use efficiency in response to the 75% increase in atmospheric CO2 concentration employed in the study; and since the earlier work of Idso et al. (1993) had demonstrated that there was very little difference in leaf stomatal conductance between the two CO2 treatments, nearly all of this water use efficiency increase had to have been the result of the CO2-induced increase in net primary productivity that led to the 80% increases in wood and fruit production.
That this result may be typical of trees in general is suggested by the facts that: (1) in a massive review of the pertinent scientific literature, Saxe et al. (1998) observed that "increasing numbers of experiments show a lack of stomatal sensitivity to CO2," especially when the data come "from long-term experiments on larger trees rooted directly in the ground," as may also be deduced from the work of Eamus (1996), and (2) the finding of Feng (1999) that for 23 sets of trees scattered throughout western North America, the average stable-carbon-isotope-derived increase in intrinsic water use efficiency (iWUE) that occurred in response to the historical increase in the air's CO2 concentration that was experienced over the period 1800-1985 yielded essentially the same value of delta iWUE/ CO2 as that derived from the sour orange tree study of Leavitt et al. In addition, even greater natural CO2-induced increases in iWUE have been documented in various trees in Europe: by Bert et al. (1997) in the case of white fir, and by Hemming (1998) in the cases of beech, oak and pine.
Perhaps the most convincing evidence of all for the validity of the final long-term equilibrium response of the sour orange trees to the 75% increase in the air's CO2 concentration in the CO2-enriched chambers came from the actual harvesting of the sour orange trees after 17 years of study at the ultimate conclusion of the experiment in January of 2005, as described by Kimball et al. (2007), when it was determined that (1) the final CO2-enriched/ambient-treatment biomass ratio of all of the above-ground parts of the trees (other than fruit) was 1.69, and that (2) "the cumulative amount of biomass due to fruit production over the duration of the experiment was increased 85% due to elevated CO2," as also described by Kimball et al. (2007). In addition, they reported that "the vitamin C content of the fruit was increased 7% based on samples taken from the fourth through the 12th years of the experiment," citing the study of Idso et al. (2002). Consequently, not only were there many more oranges produced by the trees in the CO2-enriched chambers, many more better-quality oranges were produced.
In addition, Idso et al. (2001) discovered three soluble proteins in the leaves of the sour orange trees whose synthesis and transference from second- to first-year leaves in the early springtime of each year were influenced by the atmosphere's CO2 concentration in ways that were likely to facilitate the trees' large positive photosynthetic and biomass responses to atmospheric CO2 enrichment. Specifically, they found that the proteins appeared to function as vacuolar storage proteins, which may supply each year's first flush of new foliage with the large amounts of nitrogen needed to sustain the ultra-enhanced spring branch growth of the CO2-enriched trees, which was four to six times more rapid than that of the trees growing in ambient air in their experiment (Idso et al., 2000), and which likely provided the yearly initial impetus for the 70-80% long-term growth enhancement of the trees that was maintained throughout the remainder of each year of the last half of their long-term study as a result of the experimentally-induced 75% increase in atmospheric CO2 concentration.
References
Eamus, D. 1996. Responses of field grown trees to CO2 enrichment. Commonwealth Forestry Review 75: 39-47.
Feng, X. 1999. Trends in intrinsic water-use efficiency of natural trees for the past 100-200 years: A response to atmospheric CO2 concentration. Geochimica et Cosmochimica Acta 63: 1891-1903.
Idso, C.D., Idso, S.B., Kimball, B.A., Park, H.-S., Hoober, J.K. and Balling Jr., R.C. 2000. Ultra-enhanced spring branch growth in CO2-enriched trees: Can it alter the phase of the atmosphere's seasonal CO2 cycle? Environmental and Experimental Botany 43: 91-100.
Idso, K.E., Hoober, J.K., Idso, S.B., Wall, G.W. and Kimball, B.A. 2001. Atmospheric CO2 enrichment influences the synthesis and mobilization of putative vacuolar storage proteins in sour orange tree leaves. Environmental and Experimental Botany 48: 199-211.
Idso, S.B. and Kimball, B.A. 2001. CO2 enrichment of sour orange trees: 13 years and counting. Environmental and Experimental Botany 46: 147-153.
Idso, S.B., Kimball, B.A., Akin, D.E. and Kridler, J. 1993. A general relationship between CO2-induced reductions in stomatal conductance and concomitant increases in foliage temperature. Environmental and Experimental Botany 33: 443-446.
Idso, S.B., Kimball, B.A., Shaw, P.E., Widmer, W., Vanderslice, J.T., Higgs, D.J., Montanari, A. and Clark, W.D. 2002. The effect of elevated atmospheric CO2 on the vitamin C concentration of (sour) orange juice. Agriculture, Ecosystems and Environment 90: 1-7.
Kimball, B.A., Idso, S.B., Johnson, S. and Rillig, M.C. 2007. Seventeen years of carbon dioxide enrichment of sour orange trees: final results. Global Change Biology 13: 2171-2183.
Leavitt, S.W., Idso, S.B., Kimball, B.A., Burns, J.M., Sinha, A. and Stott, L. 2003. The effect of long-term atmospheric CO2 enrichment on the intrinsic water-use efficiency of sour orange trees. Chemosphere 50: 217-222.
Saxe, H., Ellsworth, D.S. and Heath, J. 1998. Tree and forest functioning in an enriched CO2 atmosphere. New Phytologist 139: 395-436.
Last updated 29 October 2014



